In October of 1939 Gerhard Domagk sat in a Gestapo jail. Questioning and pressuring him for days, the Nazi party wanted Domagk to write a letter refusing the Nobel Prize in Physiology or Medicine that had been awarded to him. Two German physicists who had also been jointly awarded a Nobel Prize that year had written a letter refusing the award, but Domagk refused to comply. During his two weeks in jail, Domagk’s passport was taken from him, and an official statement was made that Domagk and the physicists needed no award from outside of Germany, and would not accept their Nobel prizes. Then Domagk was released to return to work.

Bio-Domagk Gerhard Domagk accepted the Nobel Prize in Medicine or Physiology in 1947, although he was awarded it in 1939, for the development of the antibiotic sulfanilamide. From the Nobel Institute.
He worked for IG Farbenindustrie, a government-organized collaborative of companies, of which Bayer had become a part. Gerhard Domagk, with his medical degree and years of experience in chemistry and pathology, had worked there since 1927, seeking pharmaceutical remedies for bacterial infections. He was part of a school of thought that believed chemical dies could be attached to other molecules, and thus used to target bacteria inside the body.
This form of treatment was known as chemotherapy. Domagk oversaw a team of chemists who systematically synthesized likely chemicals, and Domagk and his technicians treated lab animals infected with deadly bacteria or protozoa with those chemicals.
They had been screening chemicals in this way for years, and in 1931 had discovered that Atabrine worked as an anti-malarial. In 1933 they found a combination that worked against Streptococcus and Staphylococcus. He named the drug Prontosil. It was active in live animals but not in cultured cells, leading other researchers to believe it was actually a metabolite of Prontosil that was the key ingredient. They were correct, and both Prontosil and its metabolite sulfanilamide were shown to be clinically effective. Sulfanilamide is not very soluble in water, so it was frequently applied as a powder for external purposes.
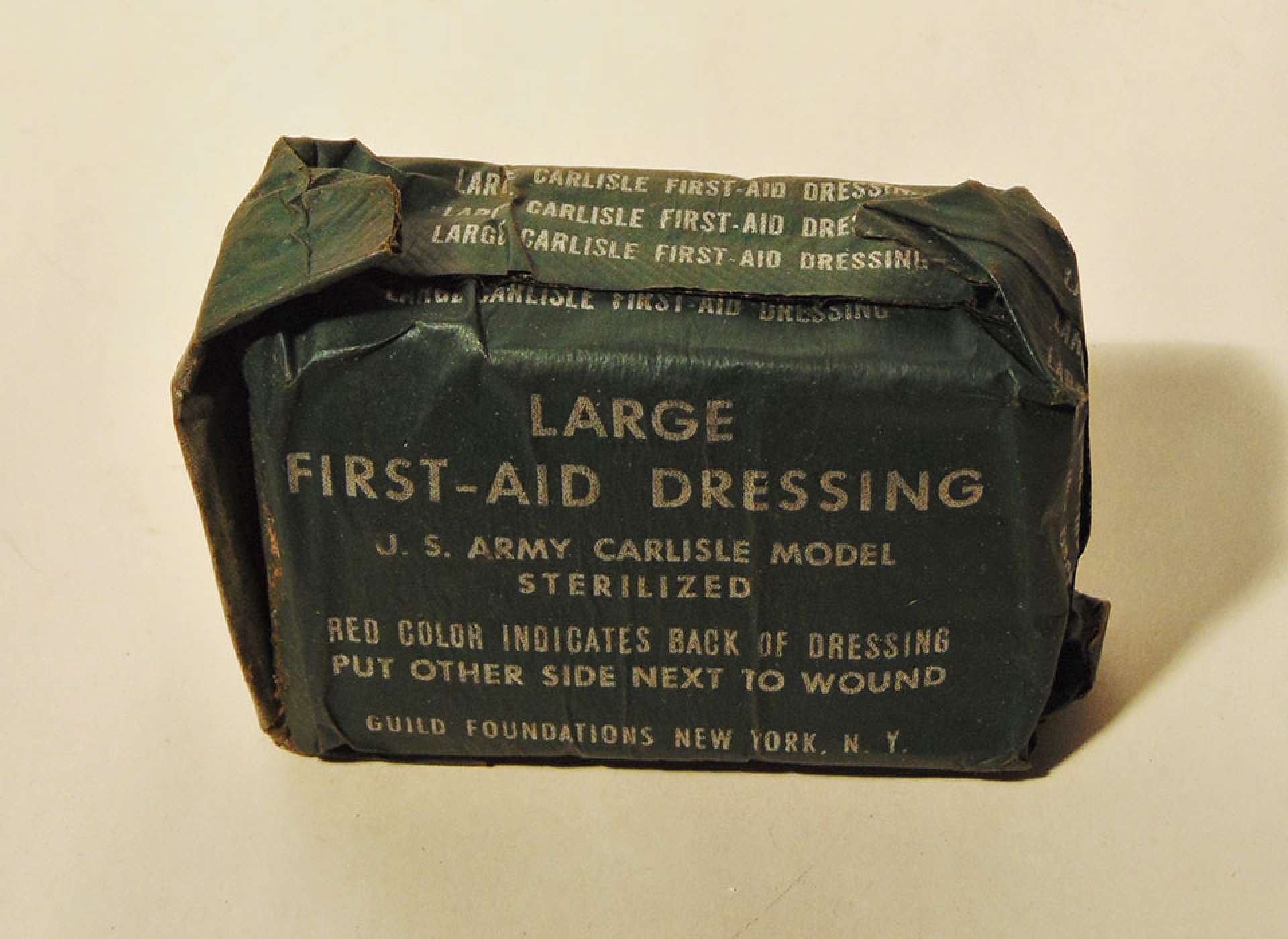
The Carlisle kit was a bandage package in a pouch carried by all soldiers on their belt. The bandage was coated with sulfanilamide. From the collection of The National Liberation Museum, Netherlands, #094.117.
If you watch a World War II movie like Band of Brothers, you’ll see medics sprinkling a yellow powder on wounds—that’s sulfa powder, or sulfanilimade. The ubiquitous bandage packs given to soldiers in the war years were coated in it. By 1939, when Domagk was in Gestapo detention, it was used worldwide. In fact, Franklin Roosevelt Jr. was prescribed it in 1936 to treat a sinus infection, and Winston Churchill used it in 1943 for a case of pneumonia. However, IG Farbenindustrie was disappointed because sulfanilamide was a common, and unpatentable, product.
Gerhard Domagk was a medical student when he left school to serve in World War I. The bloody surgeon’s tents where he worked as a medic convinced him of the need to find medicines to combat infection. When he returned to his medical studies, he was strongly influenced in the chemotherapy tradition.
Another medical approach to infections also found its origins in World War I. Some British physicians set up research camps in tents outside of field hospitals in France. They were searching for a way to vaccinate patients against infection. What their research taught them was that there were many more types of bacteria than they knew, making a vaccine irrelevant.
That did, however, set off a new direction in medical research. British medical researchers focused on types and bacteria, their characteristics, and methods to culture and identify them. One of these researchers was Alexander Fleming. After serving in those research and field hospital tents in France, he returned to his post at St Mary’s Hospital in London, where he worked on growing and identifying strains and characteristics of Staphylococcus. In 1928 he discovered a fungus growing as a contaminant on his plates. Where the fungus grew, the bacteria could not. He investigated this, and even tested some extracts of the fungus on infected mice, with positive results. He reported his research in scientific papers in 1929, but could not isolate enough of the active chemical in the fungus, called Penicillium, for mass production.
Using Fleming’s paper, in 1940 a research group at Oxford University was working to cultivate the fungus and isolate the active compound. Spurred by the onset of war, they were searching for useful treatments. They used chemical extracts to inhibit bacterial growth in both cultures and in infected mice. Unable to develop a means to produce a useful quantity of the potentially life-saving drug, the head of the research team, Howard Florey, went to the US to seek help in its development.

Howard Florey led the Oxford University research team that brought penicillin to the US for development as an antibiotic drug. From the Nobel Institute.
Working first with Merck, they tested a potential drug on a patient with sepsis. The treatment was successful, but used half of the entire drug supply they had produced. Three months later they had produced enough for 10 patients—progress was slow. A year before D-Day, the War Production Board laid out a plan to provide enough penicillin for all Allied troops. The plan identified 4 main steps to solving the problem: 1) Finding a strain of the fungus that produced a higher quantity of penicillin; 2) Finding a process to grow large quantities of the fungus; 3). Finding an efficient method of extracting the penicillin from the fungus and it’s growth medium; and 4) Packaging the penicillin in consistent doses for use.

A technician from Merck purifying penicillin in 1942. From the Imperial War Museum 43467, in the public domain.
They found a cantaloupe in a lunchroom at the Northern Regional Research Laboratory (an agricultural research center) with a very potent Penicillium growing on it. Chemical engineer Margaret Hutchinson Rousseau developed a fermentation plant to grow large quantities of the fungus. Pfizer participated in the packaging and dosing. Doses were available for the D-Day invasion, saving an estimated 15% of the wounded from death or amputation.
Fleming and Florey never patented penicillin, believing that would be unethical. They shared the Nobel Prize for Physiology or Medicine in 1945 with one other colleague, Ernst Chain. In 1947 Gerhard Domagk and his wife were able to attend the Nobel Prize ceremony, and Domagk collected his medal. He continued his research after the war, finding an effective treatment for tuberculosis.
Further work advanced the use of penicillin. In the late 1940s another chemical was added into the drug to reduce its excretion, thus reducing the frequency of dosages required. In 1945 Dorothy Crowfoot Hodgkin, of Oxford University, elucidated the structure of penicillin using X-Ray Crystallography. This allowed the production of closely related drugs.

A physical model of the penicillin molecule, constructed based on the x-ray crystallography of Dorothy Crowfoot Hodgkin. From the Science Museum of London.
If you want to read more about this topic I suggest The Mold in Dr Florey’s Coat by Eric Lax, and The Devil Under the Microscope by Thomas Hager.
Museum Store
Support the continuing educational mission of The National WWII Museum by shopping our Museum Store book selection here.
Rob Wallace
Rob runs teacher workshops and develops curriculum, including Real World Science, funded by The Northrop Grumman Foundation.
Cite this article:
MLA Citation:
APA Citation:
Chicago Style Citation:









